|
(d) For f block elements, group number is always IIIrd /3B (n-1) d – subshell + number of electrons in valence shell (nth shell)Īlternately you can calculate group number = Number of electrons in (penultimate shell + valence shell) - 8 (c) For d – block elements, group number is equal to the number of electrons in (b) For p – block elements group number is equal to 10 + number of electrons in the valence shell. (a) For s – block elements, group number is equal to the number of valence electrons. The group of an element is predicted from the number of electrons in the valence shell or a penultimate shell (n-1) as follows The orbital containing the last electron indicates the block of an element. Principal quantum number of the valence shell corresponds to the period of an element. We can easily predict the group, period & block of any element from it’s electronic configuration by the following ways PREDICTION OF GROUP, PERIOD AND BLOCK OF A GIVEN ELEMENT Most of the elements of the actinide series are radioactive They show variable oxidation states & their compounds are generally coloured They are heavy metals with high melting & boiling points There are two series of f–block elements, which areĤf series – Lanthanides 14 elements Ce(58) to Lu(71) and 5f series Actinides 14 elements Th(90) to Lr(103). These are placed separately below the main periodic table and are mainly related to IIIB i.e. Most of the transition metals form alloys. They form both ionic & covalent compounds and compounds are generally coloured and paramagnetic in nature They show variable oxidation states and are good conductors of heat & electricity. Ionization energy is between s & p- block elements These are hard, ductile & malleable metals with high melting & boiling points There are four series of d-block elements, which are 3d series−Sc (21) to Zn (30) 4d series – Y( 39) to Cd(48) & 5d series The outermost electronic configuration is (n-1) d 1-10 ns 1-2 (n=4 to 7). 3 to 12 groups of the modern periodic table. These are present in the middle part of the periodic table (between s & p block element) and constitute IIIB to VIIB, VIII, IB & IIB i.e. 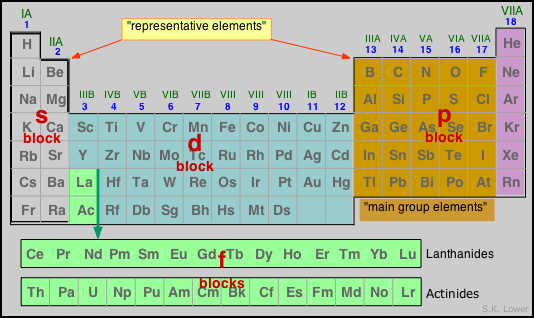
Reducing character increases from top to bottom in a group & oxidizing character increases left to right in a period. Ionization energy is higher as compared to s – block elements. The metallic character decrease from left to right along the period and metallic character increases from top to bottom within a group The last electron enters in p − orbital of valency shell and electronic configuration of valency shell is ns 2 n 1−6(n=2 to 7) Most of these elements are metalloids & non metals but some of them are metals also. group 13 to 18 of the modern periodic table. These are present in right part of the periodic table and constitute the groups IIIA to VIIA and zero groups i.e. They are strong reducing agents & are good conductor of heat & electricity. Most of the metals of this block impart characteristic color to the flame They have valency +1(in case of alkali metals) &+2 (in case of alkaline earth metals) They are highly electropositive and having low ionization enthalpies They posses metallic character & reactivity of metal increases down the group They are soft metal with low melting & boiling points Electronic configuration of valence shell is ns 1-2 These are present in the left part of the periodic table in group IA & IIA i.e. This division is based on the name of orbital which receives the valence electron.Įlements in which the last electron enters the s- orbital or their respective outermost shells are called s- block elements. On the basis of electronic configuration, the elements may be divided into four groups, that is s, p, d & f.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
